Conservation management
Managing and conserving threatened and endangered species in the wild is a difficult process. There are a large number of possible threats, outcomes, and it’s often not clear which of these (or how many of these) are at play at any one given time. Thankfully, there are also a large number of possible conservation tools that we might be able to use to protect, bolster and restore species at risk.
Using genetics in conservation
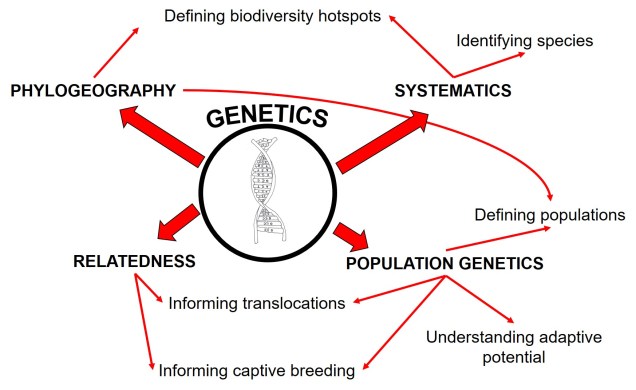
Naturally, we’re going to take a look at the more genetics-orientated aspects of conservation management. We’ve discussed many times the various angles and approaches we can take using large-scale genetic data, some of which include:
• studying the evolutionary history and adaptive potential of species
• developing breeding programs using estimates of relatedness to increase genetic diversity
• identifying and describing new species for government legislation
• identifying biodiversity hotspots and focus areas for conservation
• identifying population boundaries for effective management/translocations
This last point is a particularly interesting one, and an area of conservation research where genetics is used very often. Most definitions of a ‘population’ within a species rely on using genetic data and analysis (such as Fst) to provide a statistical value of how different groups of organisms are within said species. Ignoring some of the philosophical issues with the concept of a population versus a species due to the ‘speciation continuum’ (read more about that here), populations are often interpreted as a way to cluster the range of a species into separate units for conservation management. In fact, the most commonly referred to terms for population structure and levels are evolutionarily-significant units (ESUs), which are defined as a single genetically connected group of organisms that share an evolutionary history that is distinct from other populations; and management units (MUs), which may not have the same degree of separation but are still definably different with enough genetic data.

This can lead to a particular paradigm of conservation management: keeping everything separate and pure is ‘best practice’. The logic is that, as these different groups have evolved slightly differently from one another (although there is often a lot of grey area about ‘differently enough’), mixing these groups together is a bad idea. Particularly, this is relevant when we consider translocations (“it’s never acceptable to move an organism from one ESU into another”) and captive breeding programs (“it’s never acceptable to breed two organisms together from different ESUs”). So, why not? Why does it matter if they’re a little different?
Outbreeding depression
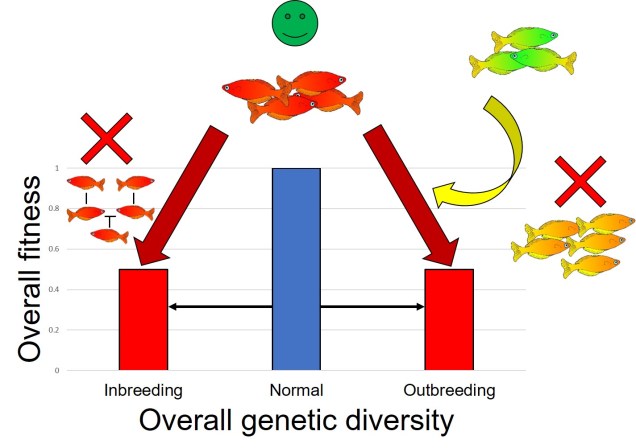
Well, the classic reasoning is based on a concept called ‘outbreeding depression’. We’ve mentioned outbreeding depression before, and it is a key concept kept in mind when developing conservation programs. The simplest explanation for outbreeding depression is that evolution, through the strict process of natural selection, has pushed particularly populations to evolve certain genetic variants for a certain selective pressure. These can vary across populations, and it may mean that populations are locally adapted to a specific set of environmental conditions, with the specific set of genetic variants that best allow them to do this.
However, when you mix in the genetic variants that have evolved in a different population, by introducing a foreign individual and allowing them to breed, you essentially ‘tarnish’ the ‘pure’ gene pool of that population with what could be very bad (maladaptive) genes. The hybrid offspring of ‘native’ and this foreign individual will be less adaptive than their ‘pure native’ counterparts, and the overall adaptiveness of the population will decrease as those new variants spread (depending on the number introduced, and how negative those variants are).

You might be familiar with inbreeding depression, which is based on the loss of genetic diversity from having too similar individuals breeding together to produce very genetically ‘weak’ offspring through inbreeding. Outbreeding depression could be thought of as the opposite extreme; breeding too different individuals introduced too many ‘bad’ alleles into the population, diluting the ‘good’ alleles.
Genetic rescue
It might sound awfully purist to only preserve the local genetic diversity, and to assume that any new variants could be bad and tarnish the gene pool. And, surprisingly enough, this is an area of great debate within conservation genetics.
The counterpart to the outbreeding depression concerns is the idea of genetic rescue. For populations with already severely depleted gene pools, lacking the genetic variation to be able to adapt to new pressures (such as contemporary climate change), the situation seems incredibly dire. One way to introduce new variation, which might be the basis of new adaptation, bringing in individuals from another population of the same species can provide the necessary genetic diversity to help that population bounce back.

The balance
So, what’s the balance between the two? Is introducing new genetic variation a bad idea, and going to lead to outbreeding depression; or a good idea, and lead to genetic rescue? Of course, many of the details surrounding the translocation of new genetic material is important: how different are the populations? How different are the environments (i.e. natural selection) between them? How well will the target population take up new individuals and genes?
Overall, however, the more recent and well-supported conclusion is that fears regarding outbreeding depression are often strongly exaggerated. Bad alleles that have been introduced into a population can be rapidly purged by natural selection, and the likelihood of a strongly maladaptive allele spreading throughout the population is unlikely. Secondly, given the lack of genetic diversity in the target population, most that need the genetic rescue are so badly maladaptive as it is (due to genetic drift and lack of available adaptive alleles) that introducing new variants is unlikely to make the situation much worse.

That said, outbreeding depression is not an entirely trivial concept and there are always limitations in genetic rescue procedures. For example, it would be considered a bad idea to mix two different species together and make hybrids, since the difference between two species, compared to two populations, can be a lot stronger and not necessarily a very ‘natural’ process (whereas populations can mix and disjoin relatively regularly).
The reality of conservation management
Conservation science is, at its core, a crisis discipline. It exists solely as an emergency response to the rapid extinction of species and loss of biodiversity across the globe. The time spent trying to evaluate the risk of outbreeding depression – instead of immediately developing genetic rescue programs – can cause species to tick over to the afterlife before we get a clear answer. Although careful consideration and analysis is a requirement of any good conservation program, preventing action due to almost paranoid fear is not a luxury endangered species can afford.