The roles of aridification and sea level changes in the diversification and persistence of freshwater fish lineages
The process of publishing science is a lengthy one – there are many rounds of revisions, assessments, and review required before a paper can be published. With that, I’m very proud to announce that the first paper from my PhD has recently been published in the journal Molecular Ecology. This paper is a collection of a lot of complex analyses, and addressing some relatively complicated biogeographical questions, so I’ve decided to provide a simplified summary here.
The study species
This work focused on one of our lab’s (MELFU) key conservation species: the southern pygmy perch, Nannoperca australis. Spread throughout southeast Australia, the southern pygmy perch is a small-bodied (<10cm long) freshwater fish generally found in low-flow creeks and wetland habitats. They’re generally a speckled grey colour, although males get some brilliant red-orange colouring in their fins during the breeding season. Previous genetic research has also shown that populations of southern pygmy perch from far eastern Victoria, Flinders Island (in the Bass Strait) and northeast Tasmania have been separated from all others for around six million years, lending to their status as a suggested independent cryptic species dubbed Nannoperca ‘flindersi’. Although not formally described, the separation of N. ‘flindersi’ and N. australis is very apparent within their genetic variation.

In the early 2000s, MELFU established a genetic-based breeding and translocation program for southern (and Yarra) pygmy perch as part of their overarching conservation management following major populations declines during the Millennium drought. Although there has been some success in this regard – with some southern pygmy perch populations re-establishing – the species is still considered Near Threatened on the IUCN Red List. Understanding how various factors, including their evolutionary history, may contribute to their current conservation status and management is important for their ongoing survival. To this end, this paper aimed to understand how past environmental changes have shaped their evolution, distribution and persistence, and how future environmental changes may similarly impact them.
The study region
This area of inquiry is particularly important given the dramatic changes in southeast Australia’s environment over geological history. Subject to a combination of tectonic changes, sea level changes, shifting water bodies and even periods of volcanism, southeast Australia is highly variable both spatially and temporally. In the current day, there is a dramatic contrast between inland (north of the Great Dividing Range) arid and semi-arid environments with the cooler and more temperate conditions along the coast. Even within the Murray-Darling Basin, the climatic gradient between highland and lowland regions has been driven adaptive differentiation within southern pygmy perch, demonstrating the impact of environmental variation in this region on their evolution.
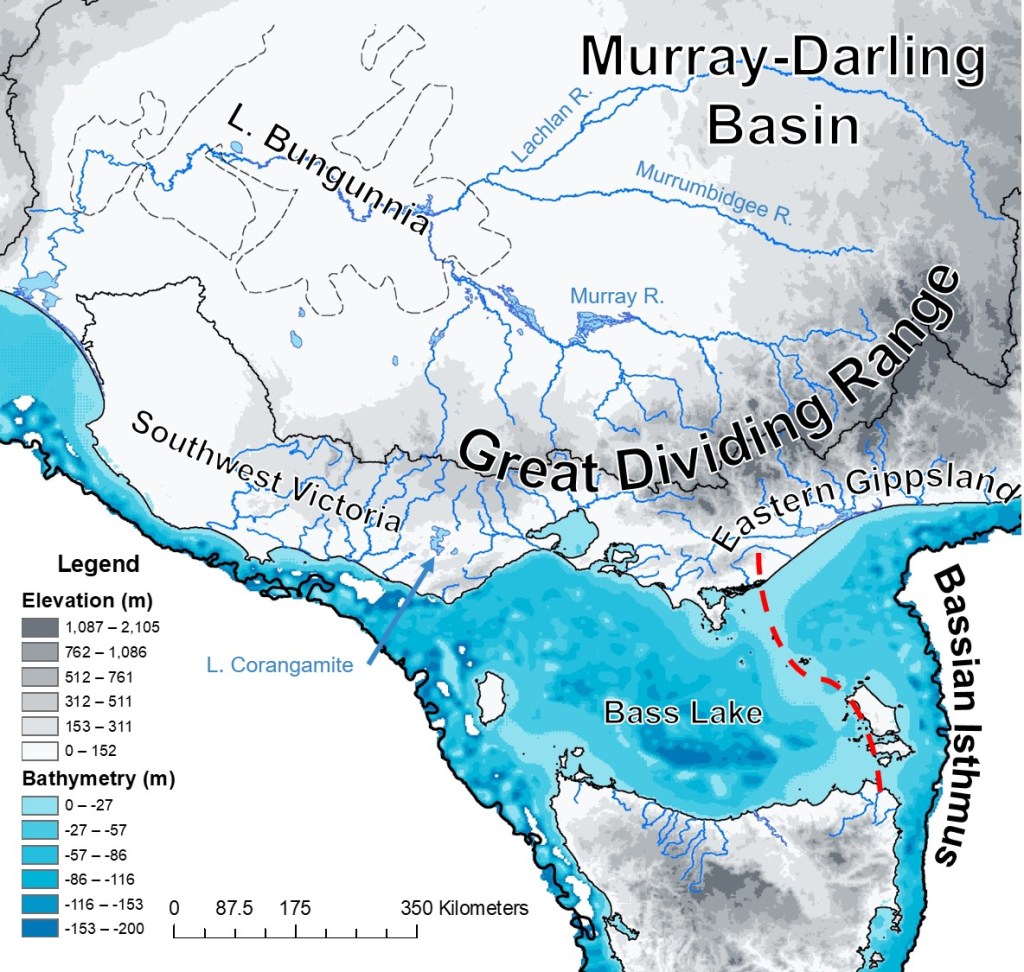
Each of these regions have also been affected by very different environmental changes over millions of years. For example, a giant paleo-lake named Lake Bungunnia once spanned across a massive area of the Murray-Darling Basin after a tectonic event created a ‘dam’ ~3 million years ago. This huge lake – spanning 90 thousand km2 at its largest size (larger than any freshwater lake in the world currently) – survived until around 700 thousand years ago, when this dam eventually burst, and the Murray River took its current shape.
However, many coastal habitats where southern pygmy perch currently occur were not connected to Lake Bungunnia. Instead, shifting sea levels in accordance with glacial cycles dramatically changed the available landscape. This included the connection of Tasmania to mainland Australia through a land-bridge called the Bassian Isthmus, which explains how they managed to travel across the “sea’ to these current islands.
Our study aims
Overall, the aims for this work were to:
- Understand the evolutionary relationships of Nannoperca australis and Nannoperca ‘flindersi’ populations.
- Understand how ancient environmental changes shaped these patterns.
- Infer the relative importance of these many environmental factors on their evolution and persistence.
To do this, we took DNA from a total of 114 individual pygmy perches – a combination of newly-collected DNA from museum samples and previous genetic work in our lab – and obtained ~8 thousand genomic fragments (ddRAD-seq loci) to analyse. We used a combination of phylogenetic, coalescent, and environmental modelling processes to get a thorough overview of these evolutionary patterns.
Our methods
Since there are a lot of methods (and this doesn’t include some things which were cut after the thesis!), I won’t go into the full detail here. To sum up, there are three main branches of analyses: phylogenetic approaches, a coalescent modelling approach, and species distribution modelling. These tackle different aspects of both the data and general study questions.
The phylogenetic approaches were used to first determine the evolutionary relationships of these populations, which was used as the basis for more thorough testing using coalescent models. We expanded upon this tree by estimating the divergence time of all branches and used an ancestral area estimation approach to suggest whether changes in distribution of these populations were ‘gradual’ (range expansion) or ‘abrupt’ (dispersal).
The bulk of the work focused on the biogeographic hypothesis testing using the coalescent. For six major splits or groups within the phylogenetic tree (see below), we devised a set of potential environmental explanations for the patterns observed. These related to different environmental changes such as sea level rise, tectonic shifts, volcanic events or other factors. We simulated genetic data under these conditions and then looked at how well each of these matches our observed genetic data, choosing the best-fitting model for each “set” as the most likely scenario.
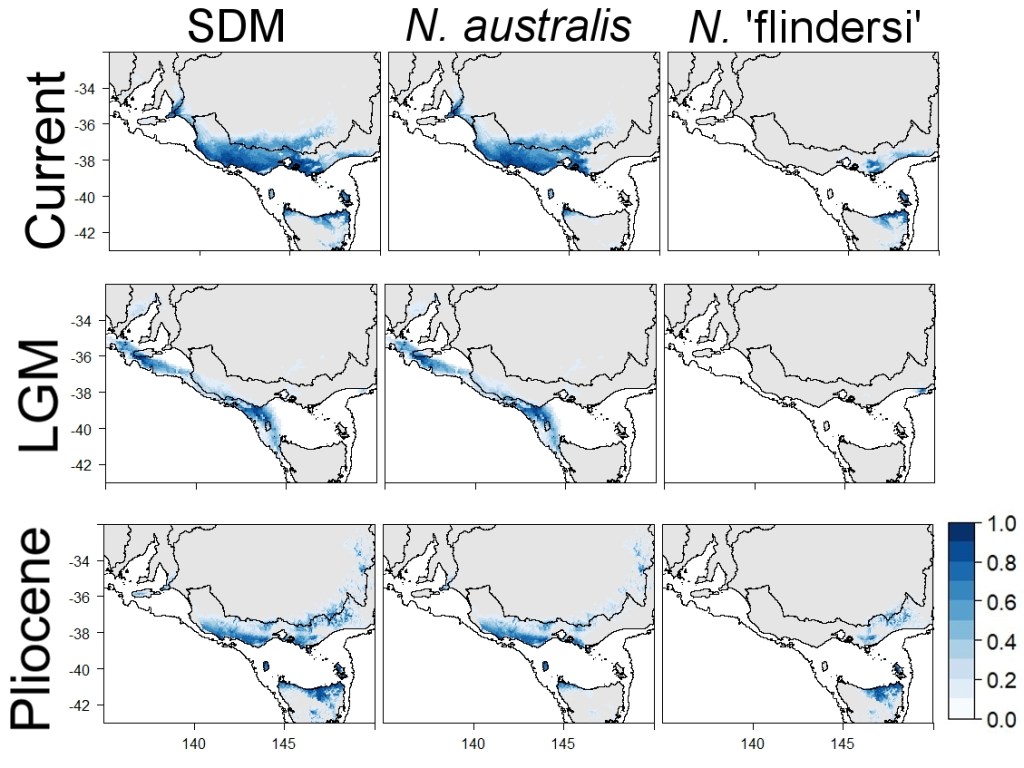
Lastly, we used a non-genetic species distribution modelling approach to understand how the total distribution of N. australis and N. ‘flindersi’ have changed since the last glacial maximum (~18 thousand years ago) and the Pliocene (~3.2 million years ago). This helps to capture distribution patterns associated with only the environment and does not rely on any of our genetic data.
Our results
We found that populations diverged in a hierarchical manner, with the oldest divergence (between N. australis and N. ‘flindersi’) occurring around six million years ago. Nannoperca australis was separated into three distinct lineages – one of just the Wilson’s Promontory (a small peninsula in Victoria) population, another of populations from the western coast of Victoria, and a third of populations from the Murray-Darling Basin. These groups, and their divergences, became the focus of the remaining analyses.

A total of 28 different hypotheses centred around 6 different sections of the tree were simulated using the coalescent. In general, models supported some post-divergence gene flow, suggesting that although these populations were isolated a long time ago they had, at some point, come back into contact and shared genetic material. Models based on shrinking lakes or sea level changes were more likely than those related to tectonic changes or other factors (such as volcanism), suggesting that these two factors were important in driving the observed evolutionary patterns. In coastal Victoria, there was an interactive effect of both – the recent (over the last 1 thousand years) shrinking of Lake Corangamite and rising sea levels after the last glacial maximum both reduced connectivity between populations.

The species distribution models suggested that, perhaps contrary to expectations, N. australis and N. ‘flindersi’ were actually more isolated during the lowered sea levels of the last glacial maximum. Separate environmental refuges in the east and west isolated the two species, likely maintaining their very divergent nature and preventing them from merging back into a single lineage during interglacial periods.
What does it all mean?
The combination of these analyses demonstrate how large-scale environmental changes can have complex impacts on the evolution and persistence of species, especially freshwater ones. Intense aridification in inland basins and sea level rise in coastal habitat both influenced the evolution of N. australis, driving the formation of new yet isolated species and lineages. Both of these factors are likely to be exacerbated under climate change – there is already some evidence that aridification in the Murray-Darling Basin is a major threat to the persistence of these threatened species. Generally, our results highlight how spatial variation in environmental changes are important to consider – not all populations of a species will be impacted by the same environmental changes in the same way. As with everything, the story is much more complicated.

One thought on “Shifting lakes, coastlines and mountains: how millions of years of environmental changes shaped the evolution of a little fish”